-
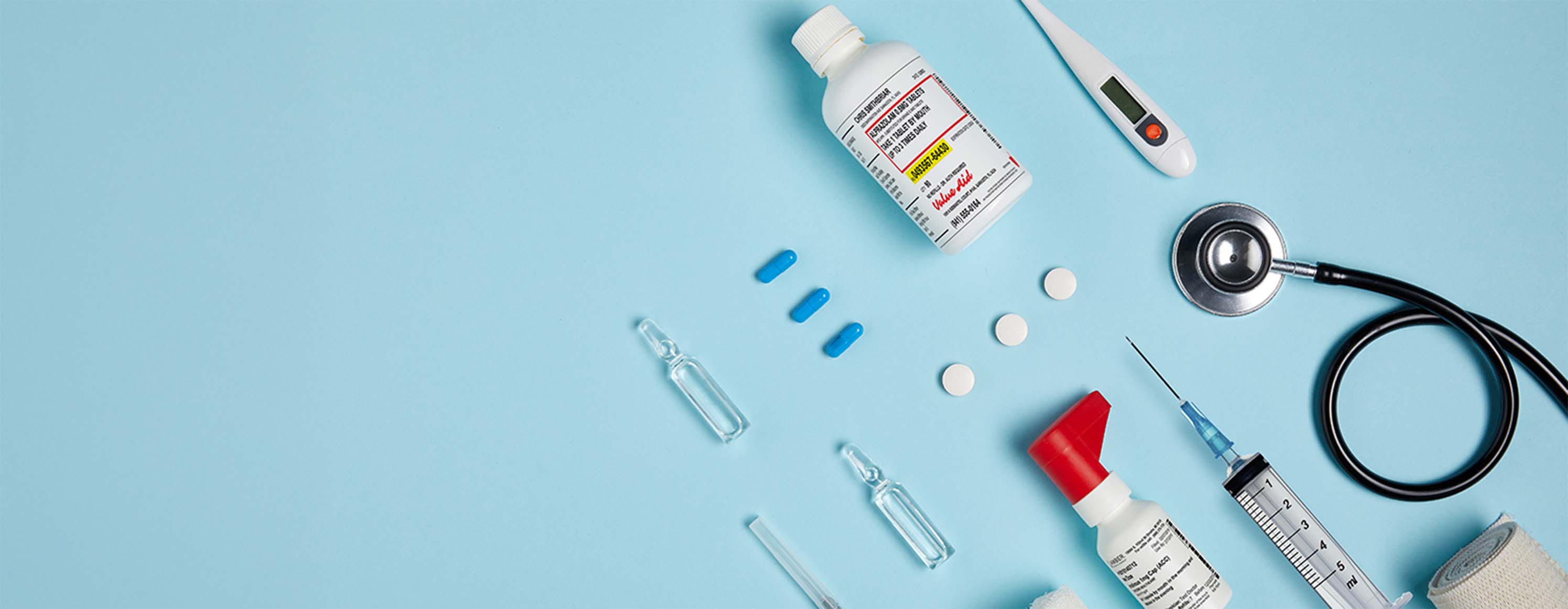
Market Commercialization
Unlock the path to market success with the BioPortUSA Commercialization Strategy.
Learn More
-

Handelsblatt GmbH Interview
BioportUSA interviewed in leading German business newspaper on the challenges German companies face when entering the US market.
-

BioPortUSA Insights: Navigating the U.S. Healthcare Market
-

BioPortUSA Speaks with Swiss Startups
BioPortUSA Joins Swissnex Boston 2023.
-

Assure Success in U.S. Markets
We identify cultural differences that exist between Europe and the USA which impacts how business is done.
-

Market Entry? We've Got You Covered.
Extensive knowledge of the Life Sciences and the US market.
-

BioPortUSA Services
The Gateway For Life Sciences Into The United States
Learn More
BioPortUSA facilitates entry for foreign-based life sciences companies into the United States. Quickly. Easily. Cost effectively.
Market Access
The United States represents a major commercial opportunity for life sciences companies and also a major challenge. BioPortUSA’s extensive knowledge of this unique environment provides strategic guidance for successful U.S. Market entry.
Legal Considerations
Operating in a regulated market requires careful consideration. Our team will guide you through the necessary steps and provide a blueprint to ensure that you have covered all the legal requirements for the U.S. Market.
FDA Requirements
FDA clearance and approval are essential if you plan to do business in the United States. BioPortUSA will provide you with a step by step strategic plan to successfully obtain FDA certification.
Build Your Brand
Marketing and branding play an important role in building visibility to your products. Our team will help you increase sales and communicate a unique message that’s appropriate for the U.S. Market.
Contact BioportUSA
Take advantage of BioPortUSA's vast industry knowledge and market experience, providing access to a menu of coordinated expert services.
CEO MESSAGE

Snapshot Of The U.S. Healthcare Market
$3.0T
U.S. Healthcare Market Value
40%
Volume of Global Sales
7200
Hospitals in The U.S. Today
$200BN
Total Sales Volume
39%
European Exports in the U.S. Life Sciences Market
<50
Employees/Company

